All You Need to Know About Clinical Research
Clinical trials are experiments designed to notice out whether an intervention is safe and effective and that the potential benefits of the intervention outweigh the risks. The intervention is nearly often a drug merely tin can be a device, such as a pacemaker or stent, or a diagnostic tool, such equally a blood exam. Participation in a clinical trial is an option for many people who have serious illnesses, especially when no proficient treatments are available. Clinical trials can also test whether treatments tin can help foreclose disorders (for example, coronary avenue illness, cancer, Alzheimer disease). Thousands of clinical trials are conducted each year and may have identify at a variety of locations, including universities, hospitals, clinics, doctors' private offices, and professional clinical research sites. Some trials also employ spider web-based or telephone communications and reporting.
The people who acquit clinical trials are called clinical researchers, or investigators. Investigators are usually doctors, but may be other health intendance professionals, who are paid to comport the trials by the National Institutes of Health (NIH) or by a pharmaceutical, biotechnologic, or medical device company. Investigators may also conduct trials supported by professional organizations such as the American Diabetes Association or the American Heart Association. Investigators follow a detailed protocol (list of instructions) that dictates who is eligible to participate in the trial, what interventions will be given or used, how often participants will be evaluated, and how information will be collected. Several thousand people typically participate in clinical trials for each new intervention before information technology becomes available to the general public.
All interventions must exist approved past the U.South. Nutrient and Drug Administration (FDA) before they can be prescribed or used (run across FDA: The Drug Evolution Process). The FDA'south goal is to let an intervention to be given to the full general public just after that intervention has proved to exist condom and constructive in carefully designed clinical trials. The FDA requires three phases of clinical trials earlier blessing is granted. An optional fourth phase is often done later an intervention is approved.
Phase I trials assess the safety of an intervention but not its usefulness in treating a illness. A phase I trial is the start fourth dimension an intervention is used on people. Tests are conducted on a small group of healthy people to acquire how the intervention acts in humans, including the side effects, and to learn what doses of drugs are safe. Considering phase I trials involve healthy people, the participants receive no direct medical benefit, simply their contribution to the health of others is significant. These participants receive monetary compensation, with the amount dependent on the time commitment and the nature of the drug or process being tested.
Phase Ii trials are conducted if the intervention seems safe in a stage I trial. In stage II trials, the intervention is tested in a larger grouping of people who have the disease that the intervention is intended to treat. Phase Ii trials help researchers determine whether the intervention is safe for sick people and give an early determination of whether the intervention is effective. If the intervention is a drug, phase II trials help researchers decide what dose might be appropriate.
Stage Three trials are conducted if safety is still satisfactory in stage II and the intervention seems effective. In phase III trials, the intervention is given to or tested in a big group of people who have the illness being studied. In phase III, the new intervention is ordinarily compared with the standard treatment, a placebo, or both.
It takes an average of 10 years from when a drug is initially discovered until it is approved for market. About 7 years of this time is for the clinical testing process. Many drugs, medical devices, and diagnostic tools never complete all 3 phases. Others do complete the 3 phases but are non approved for use because they fail to be effective or safe or both.
Stage 4 trials are conducted to evaluate interventions that take already been approved for use. I type of phase Four trial is done to compare two or more approved interventions or to test an approved intervention on a illness for which information technology is non approved. When an intervention is tested for a new disease, iii phases are not needed, simply the clinical trial design is like.
Postmarketing surveillance is another type of phase Iv trial, which is done after the drug or device has been canonical and is in widespread use in the general public. This study uses information from medical wellness records and doctors' reports to identify any side effects that were not detected in the first 3 phases of clinical trials. Postmarketing surveillance is particularly important in detecting side effects that are uncommon (and thus might not prove upwards even in a stage Three trial) or that occur mainly in a unique group of people, such equally significant women or people of a certain indigenous origin who might non accept been part of the original trials. If new side effects are identified and are serious, use of the drug or device might be restricted, or the drug or device might even exist removed from the market.
People have different reasons for wanting to participate in clinical trials. Some want the newest treatments, which they promise will be more effective than the current standard of intendance. Others participate out of a desire to contribute to scientific discipline or to receive money. Still others may want access to free drugs and medical care.
Sometimes a person's doctor recommends participation in a clinical trial. This recommendation is especially mutual for people who have cancer.
Trial recruitment ads run routinely online, in most major newspapers, and on many local radio stations. Some local newspapers and newsletters at present publish dedicated weekly sections listing clinical trials. Some trials are advertised through telephone hold messages, pecker boards, or social media sites. Many communities have one or more research centers that consumers can call directly to get information or to get on a mailing list. Almost all clinical trials are listed at www.clinicaltrials.gov, a spider web site sponsored by the NIH. Some web sites aid match people to specific trials. For example, the NIH has an net-based registry called Research Match, which people can use to connect with researchers seeking participants for their studies. CenterWatch is another service that contains thousands of trials that are currently enrolling people.
Some people find it dull to participate in a trial, especially if it lasts many months or requires frequent visits to the research site or frequent blood tests. Some trial protocols crave participants to regularly phone the trial nurse to report symptoms or to keep a medical diary at dwelling as a status of remaining in the trial.
Some trials are delayed, canceled past the sponsoring organization, or even stopped early once underway because certain participants do not fare well while taking or receiving the experimental treatment. Delays or cancellations tin be hugely disappointing to people to whom the handling brings relief. Also, later on a clinical trial has ended, participants may no longer take admission to an experimental treatment that was providing a existent benefit. Some trials are stopped early when evidence that the tested intervention is effective and safe is conclusive.
Deciding whether to participate in a clinical trial is an important and complicated decision. Both risks and benefits must exist carefully considered.
First, participants should be aware that they are not guaranteed to receive the new handling and may instead receive a placebo or older treatment.
A trial drug may have side furnishings and cause bad reactions ranging from headaches and sleeplessness to breathing difficulties or, on very rare occasions, even death. Although the researchers try to warn participants of all known side furnishings, unanticipated issues may develop.
The experimental treatment might non work as well as intended, possibly non fifty-fifty as well as standard treatment.
There are also some very real benefits to clinical trial participation. If a treatment works as expected, participants could have a better issue than with other treatments usually available to them. In some instances, participants have even been cured.
Volunteers typically receive fantabulous care in a mode that might otherwise cost thousands of dollars. Because participants are so well monitored, they tend to learn a great bargain about their overall health and any underlying medical weather. Sometimes there is an opportunity to build camaraderie with other participants, which may be specially welcome by people who take rare or uncommon diseases. At a minimum, clinical trial participants tin can exist sure that they are helping to accelerate medical scientific discipline and public health.
In a very small fraction of situations, investigators in charge of clinical trials and studies have acted unethically. One particularly shameful example is known equally the Tuskegee experiments. Conducted around Tuskegee, Alabama, from 1932 to 1972, this study enrolled about 400 poor, mostly illiterate, Black sharecroppers who had syphilis Syphilis Syphilis is a sexually transmitted disease acquired by the bacteria Treponema pallidum. Syphilis can occur in three stages of symptoms, separated by periods of apparent expert wellness. Information technology... read more 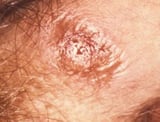 . These participants were not told that they had syphilis, and despite the widespread availability of the effective treatment penicillin, the Tuskegee investigators withheld penicillin and data nearly penicillin purely to continue to study how the affliction progressed. Participants were also prevented from accessing syphilis handling programs that were available to other people in the area. As a result of this horrendous breach of ethics and trust, several safeguards were put in place. Included in these safeguards are the institution of institutional review boards and the concept of informed consent.
. These participants were not told that they had syphilis, and despite the widespread availability of the effective treatment penicillin, the Tuskegee investigators withheld penicillin and data nearly penicillin purely to continue to study how the affliction progressed. Participants were also prevented from accessing syphilis handling programs that were available to other people in the area. As a result of this horrendous breach of ethics and trust, several safeguards were put in place. Included in these safeguards are the institution of institutional review boards and the concept of informed consent.
Institutional review boards (IRBs) are specific committees in each medical institution that participate in clinical trials. Committee members must include at to the lowest degree i non-scientific member and at least one fellow member who is not affiliated with the institution at which the research is being done. IRBs review all proposed clinical trials involving humans. The purpose of these committees is to ensure that trials are conducted in an ethical way and to avoid any unreasonable risks associated with the trial design. Merely trials that have been approved by the IRB of an institution are allowed to proceed in that institution.
Each institution's IRB may evaluate and approve studies independently from other IRBs. Alternatively, when many different institutions participate in a study, the FDA permits use of a central IRB that performs the review for that written report. Using a central IRB tin can eliminate a lot of duplicated effort by separate committees and also ensure a consequent gear up of protections and requirements at all the study centers. The cardinal IRB for a study may exist the individual IRB at 1 of the study sites or a private, independent IRB. Central IRBs must still follow all local requirements at each participating site.
Informed consent ways that a person is given all the information needed to make an educated and informed decision equally to whether to participate in a clinical trial. Information should describe all aspects of the trial, from its purpose to a argument about who pays for medical care to treat any research-related injuries. Informed consent documents tend to be lengthy (in some cases, dozens of pages long), technical, and hard to read. However, information technology is essential that participants read the documents carefully.
Participants should take the informed consent documents home, read them over several times, and discuss them with their personal doc and family members. The doctor can help clarify some of the participation risks. Family members and friends particularly need to be involved if they will exist providing transportation to the research heart. After reviewing the informed consent documents carefully, participants should return to the investigator and trial coordinator and inquire whatever further questions.
In many large clinical trials, a group of experts who are not involved in the trial and get no personal benefit related to the outcome of the trial class a data safety and monitoring board. The board regularly receives unblinded information from the trial to monitor safety and treatment efficacy. This group can recommend that the trial be stopped if results suggest higher risks to report participants than expected.
The protection of the safe and rights of clinical trial participants is a job shared by several authorities agencies too as the institutional review boards. However, to a large extent, participants—with the help of their doctor, family, and friends—must play an active office in their ain protection. The Clinical Trial Participant's Pecker of Rights can help people sympathise how to protect their rights during participation.
Clinical trial participants can always quit a trial if it proves to be uncomfortable or also inconvenient. In addition, a vigilant investigator and trial coordinator will insist that participants driblet out if in that location are changes in their health—such equally an allergic or strongly negative reaction to the trial intervention—that make the trial too risky for them to proceed. Investigators may also stop a trial if participants in one of the groups seem to be having very positive or very negative outcomes compared with participants in the other grouping. For instance, if the trial intervention is proving to exist very effective, the trial may exist stopped so that all participants may receive the intervention and benefit. If the trial intervention is proving to be ineffective or harmful, the trial may be stopped and so that no more of the participants are harmed.
The following English language-linguistic communication resource may be useful. Delight note that THE Transmission is non responsible for the content of these resources.
-
The Center for Information and Study on Clinical Inquiry Participation (CISCRP): Information for the public, patients, and other stakeholders in the clinical inquiry procedure about CISCRP'due south media outreach and awareness campaigns and live educational events; report volunteer appreciation programs; patient advisory boards and custom research assessing patient wellness journeys and study volunteer experiences; and plain language clinical trial results summaries
Listings of clinical trials are available at:
brownalearright42.blogspot.com
Source: https://www.msdmanuals.com/home/special-subjects/the-science-of-medicine-and-clinical-trials/what-participants-need-to-know-about-clinical-trials
0 Response to "All You Need to Know About Clinical Research"
Post a Comment